Let us discuss in some more detail the interaction between nucleons.
In the past there has been a tremendous experimental effort devoted
to scattering protons on protons and neutrons on protons. Since the
neutron target is not available, the neutron-neutron scattering was
inferred mostly from the scattering of protons on deuterons. All this
effort lead to a large database of cross-sections and phase shifts
that provide the most extensive information on the binary
interactions on nucleons. There have also been numerous attempts to
model the interaction between nucleons by different kinds of
potentials. Here we limit the discussion to the Argonne v![]() potential Wir95, and refer the reader to this paper for
references to other existing approaches.
potential Wir95, and refer the reader to this paper for
references to other existing approaches.
The Argonne v![]() NN interaction consists of
the electromagnetic
NN interaction consists of
the electromagnetic
![]() , one-pion-exchange
, one-pion-exchange ![]() , and
intermediate and short-range phenomenological
, and
intermediate and short-range phenomenological
![]() parts, i.e.,
parts, i.e.,
The Argonne v![]() potential adopts the point of view that at large
distances the NN interaction is governed by the OPE
effects, while the short-range part is treated fully
phenomenologically. In this respect, there is a perfect analogy
between the strong force acting between nucleons, as modelled by
Argonne v
potential adopts the point of view that at large
distances the NN interaction is governed by the OPE
effects, while the short-range part is treated fully
phenomenologically. In this respect, there is a perfect analogy
between the strong force acting between nucleons, as modelled by
Argonne v![]() , and the electromagnetic force acting between neutral
non-polar molecules, modelled by the Lennard-Jones potential.
, and the electromagnetic force acting between neutral
non-polar molecules, modelled by the Lennard-Jones potential.
Nucleons are colorless objects, i.e., when looked upon from the
outside; no net color charge is visible. The same is true for neutral
non-polar molecules that contain equal amounts of positive and
negative electromagnetic charges distributed with no net shift, and
hence they have no net charge or dipole moment. However, when two
molecules approach one another, the charges become polarized, and
each molecule acquires a non-zero dipole moment. Then the leading-order
interaction energy between molecules equals
![]() =
=
![]() , where
, where
![]() is the average electric field felt by one of the
molecules when the second one is located at
is the average electric field felt by one of the
molecules when the second one is located at
![]() , and
, and
![]() is its dipole moment. Assuming that the induced
dipole moment
is its dipole moment. Assuming that the induced
dipole moment
![]() depends linearly on the electric
field, and knowing that the electric field created by a dipole
decreases as
depends linearly on the electric
field, and knowing that the electric field created by a dipole
decreases as ![]() , we obtain immediately that
, we obtain immediately that
![]()
![]()
![]() , which gives the well-known Van der Waals
potential. At intermediate and small distances, polarization effects
become stronger, and higher induced multipole moments begin to be
active, however, we can model these effects by a phenomenological
term that is equal to the square of the Van der Waals term. Together, one
obtains the Lennard-Jones potential,
, which gives the well-known Van der Waals
potential. At intermediate and small distances, polarization effects
become stronger, and higher induced multipole moments begin to be
active, however, we can model these effects by a phenomenological
term that is equal to the square of the Van der Waals term. Together, one
obtains the Lennard-Jones potential,
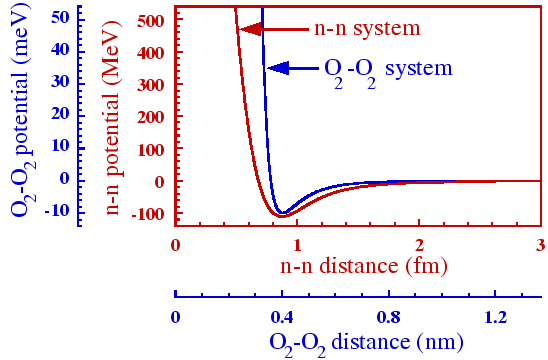 |
In Fig. 5 we show a comparison of the ![]() -
-![]() Argonne v
Argonne v![]() potential in the
potential in the ![]() S
S![]() channel, with the Lennard-Jones potential
between two O
channel, with the Lennard-Jones potential
between two O![]() molecules (
molecules (![]() =10meV and
=10meV and
![]() =0.358nm). The Argonne v
=0.358nm). The Argonne v![]() potential has been calculated by
using the av18pw.f FORTRAN subroutine Wir95, available
at http://www.phy.anl.gov/theory/research/av18/av18pot.f. Both
potentials are drawn in the same Figure with two abscissas (the lower
one for O
potential has been calculated by
using the av18pw.f FORTRAN subroutine Wir95, available
at http://www.phy.anl.gov/theory/research/av18/av18pot.f. Both
potentials are drawn in the same Figure with two abscissas (the lower
one for O![]() -O
-O![]() , the upper one for
, the upper one for ![]() -
-![]() ) and two ordinates
(the left one for O
) and two ordinates
(the left one for O![]() -O
-O![]() , the right one for
, the right one for ![]() -
-![]() ). Scales an
the abscissas were fixed so as to put the minima of potentials at the
same point, and differ by a factor of about 0.5
). Scales an
the abscissas were fixed so as to put the minima of potentials at the
same point, and differ by a factor of about 0.5![]() 10
10![]() , while
scales on the ordinates differ by the factor of 10
, while
scales on the ordinates differ by the factor of 10![]() .
.
Despite the tremendous differences in scales, both potentials are
qualitatively very similar. Amazingly, it is the electromagnetic
molecule-molecule potential that it stiffer at the minimum than the
neutron-neutron ``strong'' potential. In this respect, it is fully
justified to put the word ``strong'' into quotation marks - this
potential is not strong at all! Both potentials exhibit a very strong
repulsion at short distances - the so-called hard core (the
O![]() -O
-O![]() repulsion is stronger!). At large distances, there
appears a weak attraction (the
repulsion is stronger!). At large distances, there
appears a weak attraction (the ![]() -
-![]() attraction vanishes more
slowly - despite the exponential form of the OPE potential). Neither
of the potentials is strong enough to bind the constituents into a
composite object.
attraction vanishes more
slowly - despite the exponential form of the OPE potential). Neither
of the potentials is strong enough to bind the constituents into a
composite object.
The analogy between the ``strong'' NN force and the electromagnetic
molecule-molecule force is extremely instructive. First of all, we
can demystify the OPE potential in the sense that the exchange of
real particles (pions) is, in fact, not its essential element.
The OPE potential is a remnant of our tool (quantum field theory) that we
used to derive it, but on a deeper level it is an effect of the color
force between color-polarized composite particles. After all, nobody
wants to interpret the dipole-dipole inter-molecular O![]() -O
-O![]() force by an
exchange of a ``particle''. This force can be understood in terms of
a more fundamental interaction - the Coulomb force. Second, although
the asymptotic, large-distance, leading-order behaviour of both
potentials can fairly easily be derived, at intermediate and small
distances the interaction becomes very complicated. This is not a
reflection of complications on the level of fundamental forces (color
or electromagnetic), but a reflection of the complicated polarization
effects that take place when composite objects are put close to one
another. Moreover, these polarization effects have per se
quantum character, because the fermionic constituents do not like
being put close to one another - the Pauli exclusion principle
creates additional polarization and repulsion effects. And third, it
is obvious that at small distances there must appear effects that are
of a three-body character. Namely, when three O
force by an
exchange of a ``particle''. This force can be understood in terms of
a more fundamental interaction - the Coulomb force. Second, although
the asymptotic, large-distance, leading-order behaviour of both
potentials can fairly easily be derived, at intermediate and small
distances the interaction becomes very complicated. This is not a
reflection of complications on the level of fundamental forces (color
or electromagnetic), but a reflection of the complicated polarization
effects that take place when composite objects are put close to one
another. Moreover, these polarization effects have per se
quantum character, because the fermionic constituents do not like
being put close to one another - the Pauli exclusion principle
creates additional polarization and repulsion effects. And third, it
is obvious that at small distances there must appear effects that are
of a three-body character. Namely, when three O![]() molecules
approach each other (e.g., in liquid oxygen), the basic assumption
that they polarize one another only in pairs does not hold. There are
certainly polarization effects that depend on explicit positions of
the three of them. Similarly, when three nucleons approach each other
within the nucleus, their quark-gluon magma becomes polarized in a
fairly complicated way, which on the level of potential energy
(total-interaction energy) reveals additional terms depending on the
three positions simultaneously; this gives the three-body NNN force.
molecules
approach each other (e.g., in liquid oxygen), the basic assumption
that they polarize one another only in pairs does not hold. There are
certainly polarization effects that depend on explicit positions of
the three of them. Similarly, when three nucleons approach each other
within the nucleus, their quark-gluon magma becomes polarized in a
fairly complicated way, which on the level of potential energy
(total-interaction energy) reveals additional terms depending on the
three positions simultaneously; this gives the three-body NNN force.